Our Stories

880 CHED's Stacey Brotzel sat down with author and historian Carolina Jakeway Roemmich to talk about our new history book and how everyone seems to have a connection to the Alex.

For years, Ashley Glover did everything she could to help her children through mental health challenges. From behavioral struggles to sleepless nights, the weight on their family was immense. But everything changed when they found…
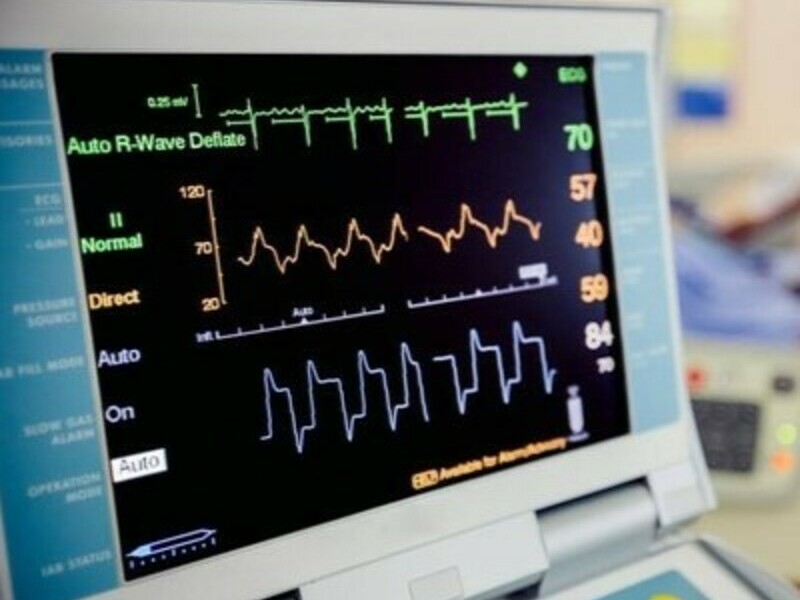
Join us in celebrating the support that has led to the acquisition of a new ECG unit for the Lois Hole Hospital for Women!

Global News kindly hosted author and historian Carolina Jakeway Roemmich and foundation CEO Sharlene Rutherford to talk about new limited-edition history book, "The Alex: The 125-Year History of the Royal Alexandra Hospital."

"The Alex: The 125-Year History of the Royal Alexandra Hospital," a sweeping and deeply human account of one of Canada’s most storied hospitals, is now available to the public.
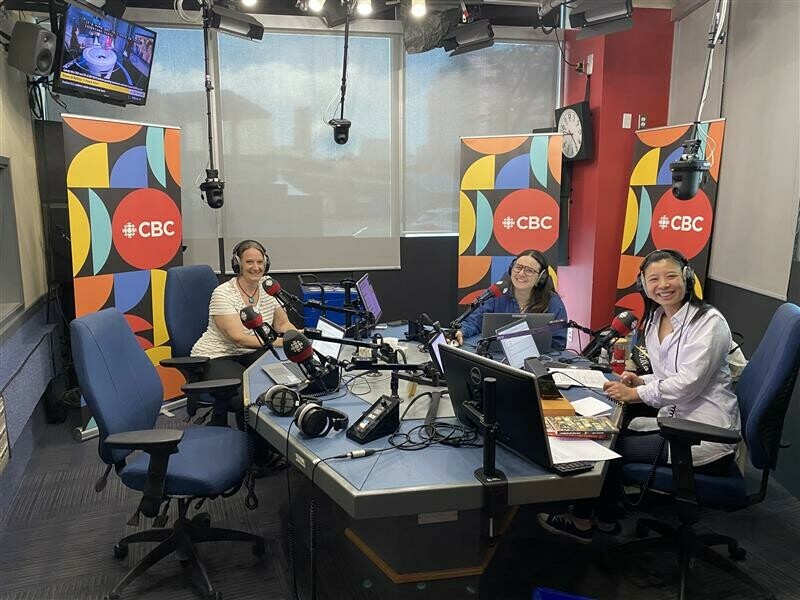
Author and historian Carolina Jakeway Roemmich sat down with CBC Radio Active to discuss our new limited-edition history book, "The Alex: The 125-Year History of the Royal Alexandra Hospital."

The Royal Alexandra Hospital Foundation was proud to once again partner with Qualico for the 2025 Links for Life Golf Invitational. We’re deeply grateful to Qualico, its generous sponsors, and all participants whose support is making…

Unit 36 gave Avery and her family extraordinary care during a time of crisis—but the space itself also needs care so it can match the life-changing work happening inside.

Authored by local historian Carolina Jakeway Roemmich, this limited-edition history book was produced by the Royal Alexandra Hospital Foundation in celebration and commemoration of our hospital’s 125-year anniversary in 2025.
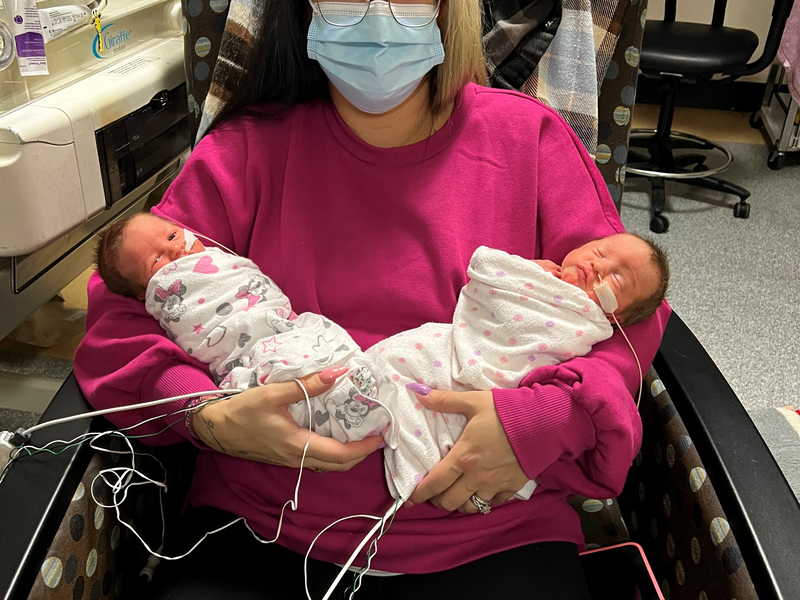
The Antepartum Recreation Program at the Lois Hole Hospital for Women supports moms navigating high-risk pregnancies while on bedrest. Learn how this program helped one mother during a time when she needed it most, in her own words.

On Saturday, May 24, 2025, Edmonton’s river valley was alive with runners, walkers, families, and friends—gathered at Sir Wilfrid Laurier Park to support women’s mental health. Now in its 13th year, the Shoppers Drug Mart…

This Vision Health Month, take time to learn about the importance of the Eye Institute of Alberta from Dr. Morely Kutzner and discover how it is changing the future of vision care through education, research, and treatment.
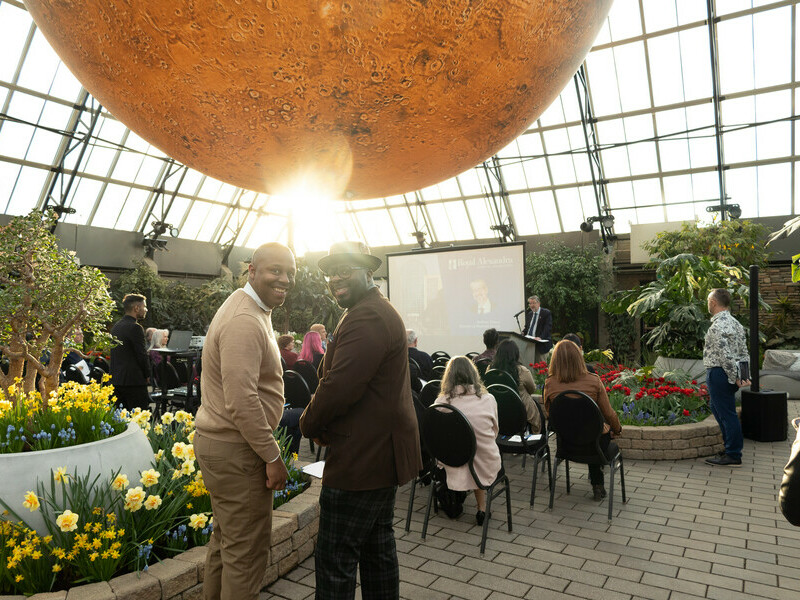
Beneath the breathtaking glass pyramids of the Muttart Conservatory, guests gathered on April 15th for an evening filled with gratitude, knowledge, and inspiration.

Joyce Helton’s lifelong love for nursing and adventure shaped a legacy that now gives back to the place where it all began.

February is Heart Month, a time dedicated to raising awareness about heart health and supporting advancements in cardiac care. During this month, we at the Royal Alexandra Hospital Foundation focus our efforts on expanding the care…

Join us in welcoming Scott Lister to the Royal Alexandra Hospital Foundation as our first-ever Vice President, Operations.

The Royal Alexandra Hospital Foundation extends its gratitude and congratulations to the Government of Alberta after it announced it will provide $1.8 million in funding to ensure the continued operations of the Bridge Healing program.

Your Heart Matters! Come partake in Listen to Your Heart at the West Edmonton Mall on February 22nd and stay informed about your heart health this Heart Month.

Full House Lottery is BACK for 2025, with an incredible suite of prizes to be won! Learn about the fantastic options available and get in on the action today!

Learn more about Heart Month at the Royal Alexandra Hospital Foundation, and how we are helping to make a difference in cardiac care this year.